TEE, transoesophageal echocardiogram ICE, intracardiac echocardiography. Practices determined by 5-digit ZIP code only those with ≥5 ablations in the study cohort were included here (n=337 providers). For each imaging modality (CT/MRI, TEE, ICE), the use of imaging is plotted on the x-axis (% of ablations where the modality is used) and the proportion of practices using the modality at the corresponding rate is on the y-axis. Practice variation in utilisation of periprocedural imaging modalities. In order to assess the validity of the study population and the robustness of the findings, we performed subsequent sensitivity analyses: (1) excluding patients with a diagnosis of atrial flutter and (2) using different follow-up periods for the endpoints (bleeding within 30 days, stroke/TIA within 90 days and repeat ablation at 1 year).Īll statistical analyses were performed using SAS software (V.9.3, SAS Institute, Cary, North Carolina, USA). We accounted for both provider variation in care delivery and clustering of patients within providers by including provider-level random intercepts in all models. In addition to indicators for periprocedural TEE, CT/MRI and ICE, all models included patient demographics, baseline characteristics, procedure year and inpatient status. We compared adjusted outcomes according to the use versus non-use of a given modality, regardless of the other imaging modalities used in a given patient. In order to test our hypotheses, we used proportional hazards regression models to estimate the association of periprocedural imaging with clinical outcomes. Data for all patients not experiencing death or repeat ablation were censored when they enrolled in Medicare managed care or at the end of the follow-up period (31 December 2009). In this study, death and repeat ablation were treated as competing risks for each other and for all other clinical and imaging events. All event and imaging rates were estimated using the cumulative incidence function, which accounts for censoring and competing risks, and differences between groups were assessed using Gray tests. Observed event rates were presented by periprocedure imaging group. Differences were assessed using Kruskal–Wallis tests for continuous variables and χ 2 tests for categorical variables. Continuous data were presented as means with SDs and categorical data as frequencies with percentages. Patients were categorised into four groups based on the combination of periprocedural TEE and CT/MRI they received. We described baseline characteristics by periprocedural imaging received. We limited practice-level results to those with five or more ablations in the study cohort. Practices were defined by 5-digit ZIP code of the physician performing the ablation. Practice-level variability in imaging utilisation was also presented using histograms. χ 2 Tests were used to assess differences within subgroups. To describe the periprocedural imaging received by the study population, we presented the number and per cent of patients receiving each imaging procedure, overall and by subgroups defined by year, age (<75, ≥75), CHADS 2 score (0–1, ≥2) and presence of heart failure. More specifically, we hypothesised that the use of periprocedural imaging would be associated with better outcomes postprocedure, including repeat ablation. 4– 6 Therefore, the objectives of the current analyses were to: (1) describe the contemporary use of periprocedural imaging technologies in a broad cohort of patients undergoing ablation for AF and (2) describe the association between periprocedural imaging use and outcomes. 
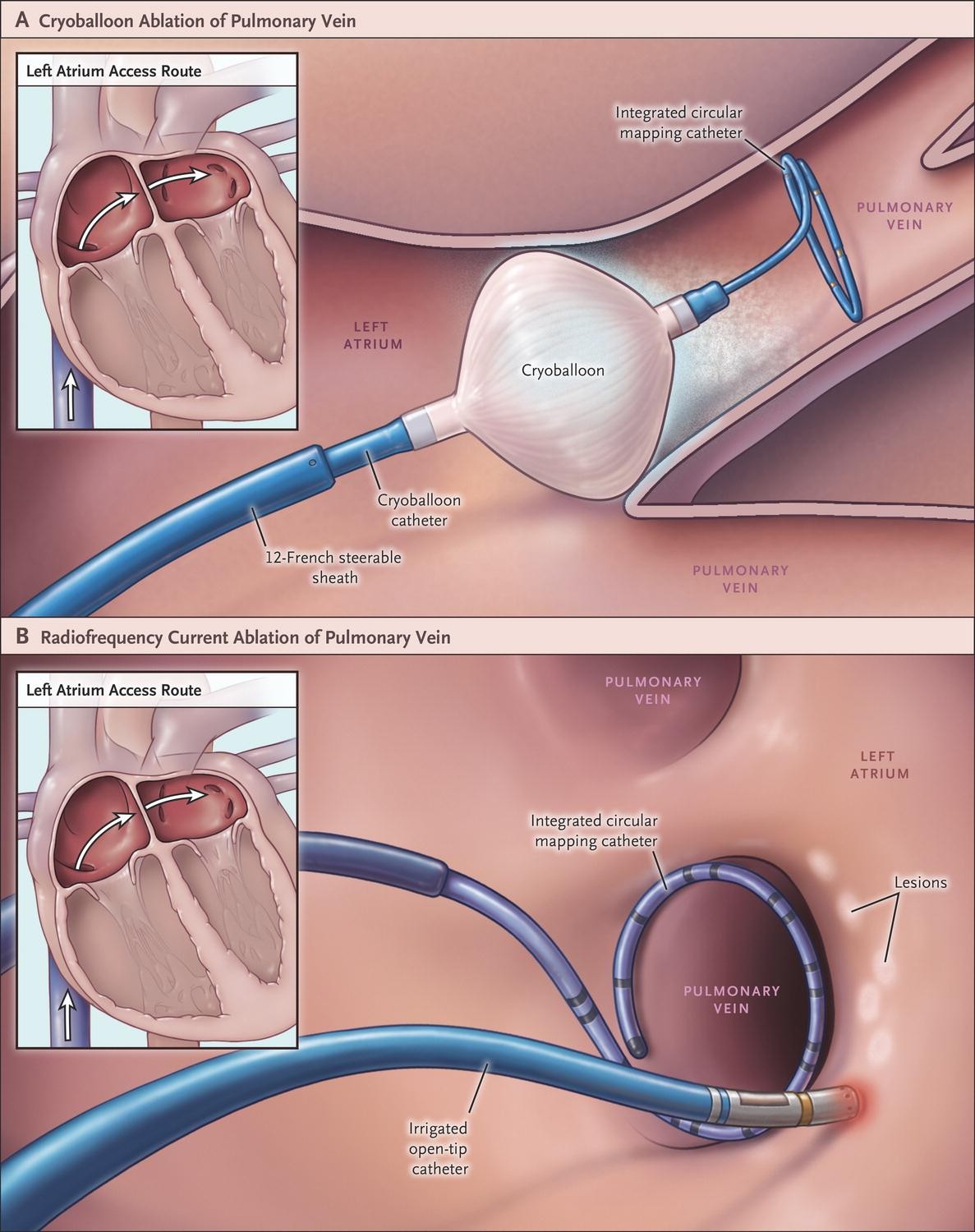
2 Furthermore, there is little evidence to demonstrate improved outcomes with any specific periprocedural imaging strategy. The extent to which adjunctive imaging is employed to guide AF ablation in practice is poorly described the largest studies include international surveys of high-volume centres that are not representative of community practice. 3 CT and/or MRI may be used to define anatomy prior to the ablation transoesophageal echocardiography (TEE) is often used to exclude the presence of left atrial appendage (LAA) thrombus and intracardiac echocardiography (ICE) and/or electroanatomic mapping (EAM) can be used for spatial guidance during the procedure. 2 In particular, cardiovascular imaging before and during AF ablation has become integral to the procedure. This may be due to a variety of factors, including better patient selection, technological advances and greater operator experience. 1 The past decade has produced significant advances in catheter ablation approaches for AF and improved procedural outcomes. Catheter ablation has been shown to be superior to antiarrhythmic therapy for the treatment of drug refractory, symptomatic atrial fibrillation (AF). 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
